From natural gas to polythene bags

Those of you who have both been following this blog closely and have a long memory will recall that in my Great Moments article about the discovery of polythene, I described its chemical make-up as “essentially a whole bunch of ethylene molecules stuck together in a chain”.
I have since received countless letters and emails* from customers, chance visitors to the blog and Nobel prizewinning chemists asking me to add some substance and hard science to that statement. Unfortunately, as the holder of a grade C ‘O’ level in Chemistry, earned more years ago than I care or am actually able to remember, I may not be the best person to take on this task.
Nevertheless, ever alert to the needs of my countless followers*, I have decided to bite the bullet and do my best to enlighten you. Please be aware, however, that readers should conduct further extensive research of their own before utilising any of the following information in an exam situation, and that neither the Davpack Blogger nor Davpack itself can be held responsible should the science contained here not comply with the standards dictated by your local examinations board.
Let’s start at the beginning
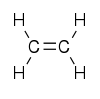
Ethylene is an organic compound, which in turn is a naturally occuring substance consisting of two or more different chemical elements that can be separated into simpler substances by chemical reactions. It’s a colourless gas with the chemical symbol C2H4 – that means it comprises two atoms of carbon and four of hydrogen. These are split into two CH2 groups connected by a double bond, making it look something like this:
Although it occurs naturally, retrieving ethylene from nature for industrial use is highly impractical, so these days it gets manufactured. This is usually done by extracting it from natural gas or oil via processes called ‘fractional distillation’ and ‘steam cracking’, although dehydrating ethanol will also do the trick.
Ethylene is used to make three fantastically useful compounds – ethylene oxide, ethylene dichloride, and ethylbenzene – which in turn are used in the manufacture of all kinds of everyday items.
The polymerisation of ethylene
The other major use – and the one of principal interest to us as packaging suppliers – is in the manufacture of polythene, or polyethylene, from which our clear polythene bags, bubble wrap and lots of other popular packaging materials are made. Polythene is a ‘polymer’, a chemical (ie one that does not occur naturally) compound or mixture of compounds made up of multiple repeating units. Polymerisation used to be carried out at very high temperatures and under pressure, but these days the process normally involves the introduction of some kind of catalyst, and in the case of polythene, that will usually be titanium(III) chloride or chromium(VI) oxide.
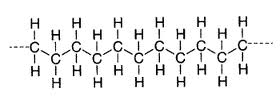
The chain is formed by breaking one of the bonds linking the carbon atoms; a new single bond can then be made with another external carbon atom, creating a link something like this:
Occasionally, one of the hydrogen atoms is replaced by a link to another carbon atom, thus creating a new chain branching off in another direction. Low Density Polythene (LDPE) has a high degree of branching, which makes it particularly suitable for the manufacture of plastic bags – its lower tensile strength and increased ductility are a result of the weaker molecular forces involved. This in turn is because (and I’m quoting here): “the instantaneous-dipole induced-dipole attraction is less”.
Davpack stocks all kinds of polythene packaging supplies. To find out more, visit our website, or call 0844 800 9844, where our friendly Sales team will be more than happy to answer any questions you might have about polybags, modern techniques in polymerisation and instantaneous-dipole induced-dipole attraction.
*as in there’s nothing to count
Dave Smith
Latest posts by Dave Smith (see all)
- An environmental guide to plastic carrier bags - 22nd June 2015
- Millimetres and litres: a guide to choosing the right sized box - 11th June 2015
- The Davpack Christmas Sale – There’s More! - 12th November 2014